
If you ask Sue Lee who her best friend is, she won’t hesitate to tell you it’s Jenny, her big sister. “Jenny was two years older than me,” says Sue. “My earliest memory is of waiting for her to come home from kindergarten. She would always save her snack and bring it home to share it with me.”
Sue grew up in Korea as the youngest of eight children, two boys and six girls. When she was 16, her family moved to Los Angeles, and she and Jenny attended the local high school together. “We didn’t speak English, so not a lot of people talked to us,” recalls Sue. “But Jenny and I didn’t care because we had each other.”

Although Sue had been blessed with wonderful siblings, they shared a tragic legacy: a strong family history of gastrointestinal (GI) cancer. Sue’s grandfather, cousin, and several of her father’s siblings had died of it — and when she was 30, her father also passed away early from colon cancer. “I knew I wanted to practice medicine, and I’d already finished residency for internal medicine when my father got sick,” says Sue. After his death, Sue changed her focus to GI. “Making that switch was a no-brainer. I think he’d be proud of me, knowing he inspired me to help others.”
After watching so many family members die from GI cancer, it never occurred to Sue that she could develop any other cancer — but she still took precautions. “My sisters would all tease me for getting an annual mammogram because I’m so flat-chested,” says Sue. “They had to eat those words later…” In 2017, during a self-breast exam, Sue found a lump and was diagnosed with HR+, HER2– node positive early breast cancer with a high risk of recurrence. “I couldn’t believe it,” she says. “Nobody in my family had ever had breast cancer.”
As Sue began treatment, her five older sisters worked together to provide her a powerful support system: “I was really sick, so they took turns coming out to be with me and my husband for two or three weeks at a time,” remembers Sue. “They were wonderful.” During one of those visits, Jenny said something that would haunt Sue in the years to come: “She said, ‘Why is it you who has to go through this? I’ve already lived a good life. I wish I could take this from you.’”
After two surgeries, chemo, and radiation, Sue was declared cancer-free in May 2018. The family’s six sisters had always taken an annual girls trip together but had missed the past year while Sue was in treatment. “When I was finally done, we all decided it was time to celebrate with a trip to Palm Springs, California,” she says. “I didn’t know it at the time, but we would never all go on a sisters trip together again.”

Just as Sue was setting out for the airport, she got a call from Jenny. “I thought she was going to tell me she was running late, but she said she was at the hospital.” The women immediately canceled their trip, and went to be with their sister.
Despite having had a completely normal physical just months earlier, Jenny was now facing an aggressive form of sarcoma, a rare form of cancer that develops in connective tissues, like bone or muscle. “There was no way anyone could have detected it,” says Sue. “Even after thirty-five years as a doctor, I couldn’t do anything to help her.” The tumor was behind Jenny’s heart and stomach, and she hadn’t experienced any noticeable symptoms until the tumor was so large it began pushing up against her organs. “When she learned about how serious this was,” remembers Sue, “Jenny laughed and said, ‘Well, I’ve never done anything half-assed.’”
As she processed what was happening, Sue’s mind immediately flashed back to the moment Jenny visited her in the hospital just months earlier: “She’d said she wanted to take the burden off me, and she did. Of course, I’m a doctor, and I knew I didn’t actually give this to her. But God, did that make me feel guilty.”
Sue spent three weeks by Jenny’s side in the hospital, a time she describes as the worst three weeks of her life. “My own cancer journey was hard,” she says, “but nothing prepared me for seeing my sister’s CT scan and realizing her cancer was terminal.”
As she sat by Jenny’s hospital bed — the same place Jenny had sat with her just months earlier — all she could feel was anger. “Just when I got better, my sister got sick,” says Sue. “When it happened to me, I’d accepted it. But I got better. And Jenny wasn’t going to get better.”
In July of 2018, Sue got a call from her oncologist, who said that her breast cancer had clinical characteristics — tumor size, grade, and stage — that made it more likely to return or spread to become metastatic breast cancer, which is incurable. Her doctor then explained there was a clinical trial for a medicine called Verzenio® (abemaciclib) that is used to treat certain types of breast cancer known as HR+, HER2– (hormone receptor positive/human epidermal growth factor receptor 2 negative) metastatic breast cancer and was now being studied in early breast cancer. She asked Sue if she would participate. “There was nothing that could help my sister with her sarcoma, and I would have given anything for extra time with her,” says Sue. “How could I possibly say no to something that could help other patients with the same disease as me?” Just as she’d used the grief from her father’s passing to begin a career helping others, Sue participated in the trial as a way to honor her sister.

Sue was among the trial participants who received Verzenio, a medicine taken orally twice a day, alongside hormone therapy. While Sue participated in the trial, Jenny continued to deteriorate; just two years after her diagnosis, Jenny passed away on March 3, 2020, six days before she would have turned 62. “I see so much sadness in my work every day, but nothing can prepare you for when it happens to you,” says Sue. “But as devastating and horrific as her death was, my own diagnosis had made me think differently about life. It helped me learn how to cope and to appreciate the time I had.”
Soon after Jenny’s death, Sue learned that the medicine she’d been receiving through the clinical trial had been approved by the FDA to treat HR+, HER2– node-positive early breast cancer that has a high risk of coming back as determined by a healthcare provider. Verzenio is given along with hormonal therapy to women and men. It is not known if Verzenio is safe and effective in children.
Diarrhea is common with Verzenio, may sometimes be severe, and may cause dehydration or infection. The most common time to develop diarrhea is during the first month of Verzenio treatment. If you develop diarrhea during treatment with Verzenio, your healthcare provider may tell you to temporarily stop taking it, stop your treatment, or decrease your dose. If you have any loose stools, start taking an antidiarrheal medicine (such as loperamide), drink more fluids, and tell your healthcare provider right away. Additional information on side effects is provided below.
“It was nice knowing that I played a small part in helping the fight against my type of early breast cancer,” she says. “I know my role was just a grain of sand on a big beach, but it made me happy.”
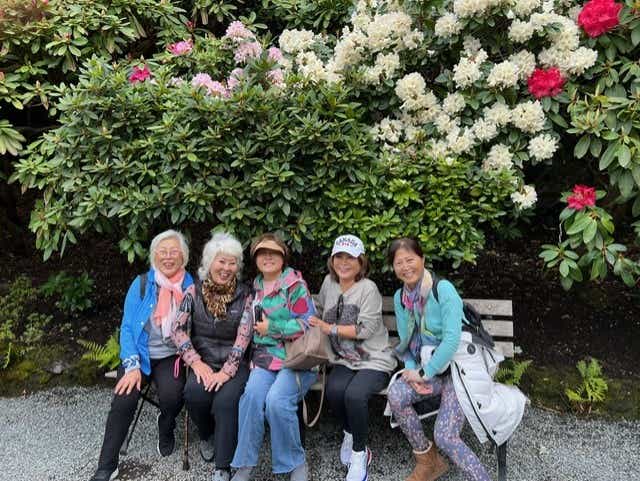
Earlier this year, Sue and her sisters went on their first sisters trip since Jenny’s passing. “Grief never leaves you, but maybe that’s OK. I never want to regret not having spent more time with the people I love.”
References
1. American Cancer Society. Breast Cancer Facts & Figures 2022-2024. Atlanta: American Cancer Society, Inc. 2022. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/2022-2024-breast-cancer-fact-figures-acs.pdf
2. Verzenio (abemaciclib). Prescribing information. Lilly USA, LLC.
3. Verzenio Patient Prescribing Information and Prescribing Information
INDICATIONS AND SAFETY SUMMARY
Verzenio® (ver-ZEN-ee-oh) is used to treat certain types of breast cancer known as HR+/HER2– (hormone receptor-positive/human epidermal growth factor receptor 2 negative) breast cancer.
It is a medicine you can take if:
- You have node-positive early breast cancer that has a high risk of coming back as determined by your healthcare provider. Verzenio is given along with hormonal therapy to women and men.
- Or, the cancer has spread to other parts of the body (metastasized). If you are female or male, then Verzenio is given with an aromatase inhibitor as initial endocrine-based therapy. If you are a female or male, and the cancer has gotten worse after hormonal therapy, then Verzenio is given with fulvestrant. If you are female or male and the cancer has gotten worse after both hormonal therapy and chemotherapy, then Verzenio is given by itself.
It is not known if Verzenio is safe and effective in children.
Warnings – Verzenio may cause serious side effects, including:
Diarrhea is common with Verzenio, may sometimes be severe and may cause dehydration or infection. The most common time to develop diarrhea is during the first month of Verzenio treatment. If you develop diarrhea during treatment with Verzenio, your healthcare provider may tell you to temporarily stop taking it, stop your treatment, or decrease your dose.
If you have any loose stools, start taking an antidiarrheal medicine (such as loperamide), drink more fluids, and tell your healthcare provider right away.
Low white blood cell counts (neutropenia) are common with Verzenio and may cause serious infections that can lead to death. Your healthcare provider should check your white blood cell counts before and during treatment. If you develop low white blood cell counts during treatment with Verzenio, your healthcare provider may tell you to temporarily stop taking it, decrease your dose, or wait before starting your next month of treatment. Tell your healthcare provider right away if you have signs and symptoms of low white blood cell counts or infections, such as fever and chills.
Verzenio may cause severe or life-threatening inflammation (swelling) of the lungs during treatment that can lead to death. If you develop lung problems during treatment with Verzenio, your healthcare provider may tell you to temporarily stop taking it, decrease your dose, or stop your treatment. Tell your healthcare provider right away if you have any new or worsening symptoms, including:
- Trouble breathing or shortness of breath
- Cough with or without mucus
- Chest pain
Verzenio can cause serious liver problems. Your healthcare provider should do blood tests to check your liver before and during treatment. If you develop liver problems during treatment with Verzenio, your healthcare provider may reduce your dose or stop your treatment. Tell your healthcare provider right away if you have any of the following signs or symptoms of liver problems:
- Feeling very tired
- Loss of appetite
- Pain on the upper right side of your stomach area (abdomen)
- Bleeding or bruising more easily than normal
Verzenio may cause blood clots in your veins, or in the arteries of your lungs. Verzenio may cause serious blood clots that have led to death. If you develop blood clots during treatment with Verzenio, your healthcare provider may tell you to temporarily stop taking it. Tell your healthcare provider right away if you have any of the following signs and symptoms of a blood clot:
- Pain or swelling in your arms or legs
- Fast breathing
- Shortness of breath
- Fast heart rate
- Chest pain
Verzenio can harm your unborn baby. Use effective birth control (contraception) during treatment and for 3 weeks after the last dose of Verzenio and do not breastfeed during treatment with Verzenio and for at least 3 weeks after your last dose. Verzenio may affect the ability of males to father a child.
Common side effects
The most common side effects of Verzenio include:
- Nausea
- Abdominal pain
- Infections
- Low red blood cell counts (anemia)
- Decreased appetite
- Headache
- Hair thinning or hair loss (alopecia)
- Tiredness
- Low white blood cell counts (leukopenia)
- Vomiting
- Low platelet counts (thrombocytopenia)
These are not all the possible side effects of Verzenio.
Tell your healthcare provider if you have any side effects. You can report side effects at 1-800-FDA-1088 or www.fda.gov/medwatch.
Before using
Before you use Verzenio, tell your healthcare provider about all your medical conditions, including:
- If you have fever, chills, or other signs of infection.
- If you have a history of blood clots in your veins.
- Have lung or breathing problems.
- Have liver or kidney problems.
- If you are pregnant, plan to become pregnant, or are breastfeeding.
- About all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you take a medicine that contains ketoconazole.
How to take
- Take Verzenio exactly as your healthcare provider tells you.
- Your healthcare provider may change your dose if needed. Do not stop taking Verzenio or change the dose without talking to your healthcare provider.
- Verzenio may be taken with or without food.
- Swallow Verzenio tablets whole. Do not chew, crush, or split the tablets before swallowing. Do not take Verzenio tablets if they are broken, cracked, or damaged.
- Take your doses of Verzenio at about the same time every day.
- If you vomit or miss a dose of Verzenio, take your next dose at your regular time. Do not take 2 doses at the same time to make up for the missed dose.
What to avoid during treatment
- Avoid taking ketoconazole during treatment with Verzenio. Tell your healthcare provider if you take a medicine that contains ketoconazole
- Avoid grapefruit and products that contain grapefruit during treatment with Verzenio. Grapefruit may increase the amount of Verzenio in your blood
Learn more
Verzenio is a prescription medicine. For more information, call 1-800-545-5979 or go to verzenio.com.
This summary provides basic information about Verzenio but does not include all information known about this medicine. Read the information that comes with your prescription each time your prescription is filled. This information does not take the place of talking with your healthcare provider. Be sure to talk to your healthcare provider about Verzenio and how to take it. Your healthcare provider is the best person to help you decide if Verzenio is right for you.
AL CON BS 03MAR2023
Verzenio® is a registered trademark owned or licensed by Eli Lilly and Company, its subsidiaries or affiliates.
PP-AL-US-4409. 03/2025. ©Lilly USA. LLC 2025. All rights reserved.









